Inflammatory Bowel Disease Long-Term Therapy UC VISIBLE 1.Ĭopyright © 2020 AGA Institute. It has a favorable safety and tolerability profile.

#XIAOPAN 2015 APK#
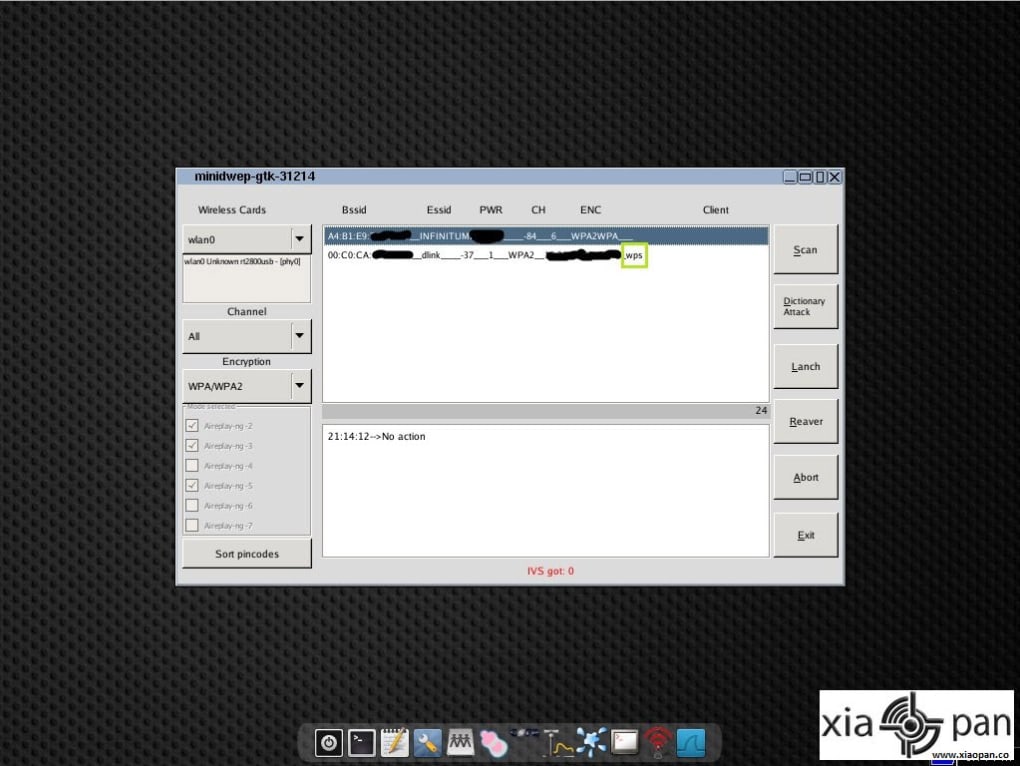
Check the hidden box to hide the apk from mobile’s app drawer. In IP section, type the hostname you have created by no-ip. Go to No-ip tab and fill your information & click on update. Subcutaneous vedolizumab is effective as maintenance therapy in patients with moderately to severely active ulcerative colitis who had a clinical response to intravenous vedolizumab induction therapy. Now extract the file, Open AndroRat Binder.exe. Subcutaneous and intravenous vedolizumab safety profiles were otherwise similar. The incidence of injection-site reactions was more frequent in patients given subcutaneous vedolizumab (10.4%) than intravenous vedolizumab (1.9%) or placebo (0%) these were not treatment limiting, most were mild, and none resulted in discontinuation. It includes a number of advanced tools for network administrators, security professionals and home users to test the strength of their wireless networks and eliminate. The subcutaneous vedolizumab group also had greater endoscopic improvement and durable clinical response at week 52 compared with placebo (both P <. How create a Xiaopan OS VM in Virtualbox Xiaopan OS is an easy to use security and penetration testing with a collection of wireless security and forensics tools. The primary end point was clinical remission at week 52, which was defined as a total Mayo score of ≤2 and no subscore >1.Īmong the randomized 216 patients, clinical remission at week 52 was achieved by 46.2%, 42.6%, and 14.3% of patients in the subcutaneous vedolizumab, intravenous vedolizumab, and placebo groups, respectively (subcutaneous vedolizumab vs placebo: Δ32.3% 95% confidence interval, 19.7%-45.0% P <.
#XIAOPAN 2015 SOFTWARE#
Xiaopan OS is an easy to use software package for beginners and experts that includes a number of advanced hacking tools to penetrate WPA / WPA2 / WPS / WEP wireless networks. At week 6, patients with clinical response were randomly assigned maintenance treatment with subcutaneous vedolizumab 108 mg every 2 weeks, intravenous vedolizumab 300 mg every 8 weeks, or placebo. Easy to use pentesting distribution for wireless security enthusiasts. Patients with moderately to severely active ulcerative colitis received open-label treatment with intravenous vedolizumab 300 mg at weeks 0 and 2.
#XIAOPAN 2015 TRIAL#
We performed a phase 3, double-blind, double-dummy trial at 141 sites in 29 countries from Decemthrough August 21, 2018. It can be trial, all you need is vmusb.sys, then you can remove it.
Make a temporary installation of version 11. Subcutaneous vedolizumab was investigated as maintenance treatment in patients with moderately to severely active ulcerative colitis. It works for me on VMWare Workstation 10.0.4, resolving the blue screen when verifier is enabled, I hope it works on version 9 too Tested on Windows 2003 圆4 SP2. Some patients might prefer a subcutaneous formulation of vedolizumab for maintenance treatment. Maintenance treatment with vedolizumab, a monoclonal antibody that inhibits the gut-selective α 4β 7 integrin, is administered intravenously.
